Elmiron
Elmiron is a prescription drug used for treating interstitial cystitis (IC) bladder pain and discomfort. IC is a chronic condition with no cure that causes bladder pain and pressure. Elmiron is designed to relieve pain by creating a barrier around the bladder wall. Lawsuits filed against Elmiron claim extended drug use has caused severe eye damage.
If you have used Elmiron and suffer from eye damage or pigmentary maculopathy, contact Counsel Hound for a no-cost consultation and case evaluation.
What is Elmiron?
Elmiron is an oral prescription drug made of pentosan polysulfate sodium. It was approved for sale in 1996 by the Orphan Drug Act which gives medication manufacturers incentives to produce treatments for rare diseases. Elmiron was created to treat the discomfort and bladder pain associated with interstitial cystitis (IC). IC is a chronic condition affecting between 3 to 8 million women and 1 to 4 million men in the United States. IC damages the lining of the bladder wall, causing urine to irritate the damaged wall.[1] There is currently no cure for interstitial cystitis, and Elmiron is the only FDA-approved medication to provide relief from the common symptoms. Elmiron is designed to create a barrier of polysulfate sodium on the bladder wall.[2]
Common symptoms of Elmiron include:[3]
- Diarrhea
- Hair loss
- Nausea
- Headache
- Stomach pain
- Dizziness
- Skin rash
Despite being the primary treatment for interstitial cystitis, studies have shown that oral polysulfate sodium may not provide much relief for many IC patients. The FDA found that after six months, less than 1.5% of patients reported pain relief.[4]
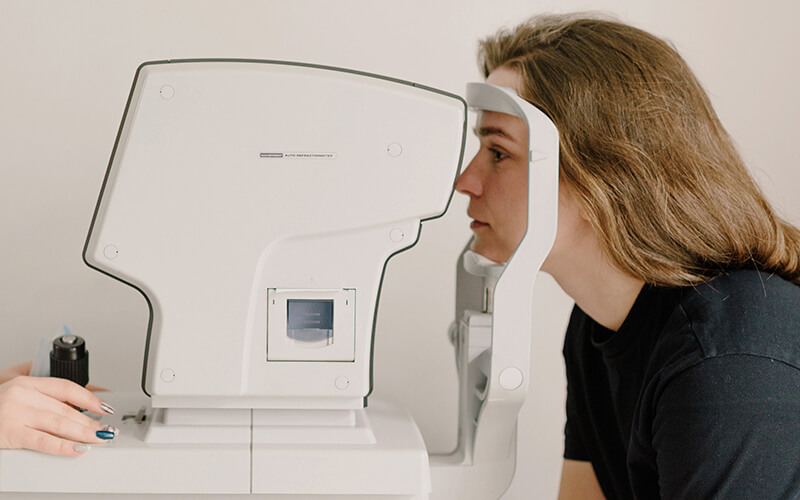
Link to Eye Damage
Studies have shown that people who take Elmiron for an extended period have an increased risk of developing pigmentary maculopathy. Maculopathy is a progressive disease of the macula that can lead to vision loss. It affects a portion of the retina called the macula. The retina is a layer of cells at the back of the eye that sends signals to the brain to form visual images. The macula is responsible for central vision and the ability to focus and see fine detail.[5]
Symptoms of Elmiron-related vision damage include:

- Blurred vision,
- Difficulty adjusting to dark lighting,
- Metamorphopsia.
Symptoms may occur as early as three years after starting Elmiron, and pigmentary maculopathy caused by Elmiron may be misdiagnosed as Age-Related Macular Degeneration (AMD).
The Elmiron medication insert did not list any warnings for eye damage until June 2020. The FDA released a new drug label that included warnings for visual impairment and pigmentary maculopathy on June 16, 2020.[6]
Compensation
Elmiron-related pigmentary maculopathy can leave you with physical, emotional, and financial hardships. Our Counsel Hound lawyers will work to maximize your settlement or award. Types of compensation you may be eligible for include economic and non-economic damages.
Economic Damages:
- Medical expenses related to your diagnosis and treatment of Elmiron-related pigmentary maculopathy.
- If your pigmentary maculopathy forced you to miss work or suffer a wage decrease at a new position, we will prove your losses with timesheets.
Non-Economic Damages:
Some damages cannot be proven with lost wages, property damages, etc., and you and your loved ones may face emotional suffering as a result of Elmiron-related vision impairment. Our lawyers will work to include all available compensation in your case.
Why file a lawsuit?
Filing an Elmiron claim can help:
- Victims of pigmentary maculopathy focus on health and treatment.
- Families afford the cost of their loved one’s medical expenses.
- Hold manufacturers and companies of Elmiron accountable.
- Encourage other victims to seek fair compensation for their injuries.
Nearly all of the cases arose from situations where troop leaders were left alone with the boys, a practice that the BSA officially prohibited in 1987.
FAQs
The amount of time you have to file an Elmiron claim depends on the statute of limitations in your state. Contact Counsel Hound to discuss your eligibility.
Counsel Hound offers no-cost consultations and case evaluations. Many lawyers work on a contingency fee meaning the attorney fees come from your settlement award once you win. If there is no recovery, you will not have to pay anything.
REFERENCES
- https://www.niddk.nih.gov/health-information/urologic-diseases/interstitial-cystitis-painful-bladder-syndrome/definition-facts
- https://cddi.ucsd.edu/Successes/drugs/elmiron.html
- https://www.rxlist.com/elmiron-side-effects-drug-center.htm
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/020193Orig1s015lbl.pdf
- https://www.drugwatch.com/elmiron/macular-degeneration/
- https://www.accessdata.fda.gov/scripts/cder/safetylabelingchanges/index.cfm?event=searchdetail.page&DrugNameID=2277
No-Cost Case Evaluation
Law is complicate matter. It can cause you a big problem if you ignore it. Let us help you!
Free Consultation
Law is complicate matter. It can cause you a big problem if you ignore it. Let us help you!

