Surgical staplers are used in hundreds of thousands of operations every year, from bariatric surgery to lung procedures. When they work correctly, they close tissue efficiently and reduce surgical time. But when they malfunction, the results can be devastating: internal bleeding, organ damage, infections, and even death. If you were injured because a surgical stapler misfired, jammed, or failed during your surgery, you may have the right to file a lawsuit against the manufacturer.
Contact Counsel Hound for a free case evaluation if you were injured by a surgical stapler. There are no fees unless you win.
This guide covers how surgical stapler injuries happen, which devices and manufacturers are involved, the types of lawsuits being filed, what compensation is available, and how to take action.
What Are Surgical Staplers?
Surgical staplers are medical devices that use rows of tiny staples to close wounds, connect tissues, or seal organs during surgery. They are widely used in procedures including:
- Bariatric (weight loss) surgery: Gastric bypass, sleeve gastrectomy, and lap-band procedures
- Colorectal surgery: Bowel resection and colon cancer treatment
- Thoracic surgery: Lung resection and lobectomy
- General surgery: Hernia repair, appendectomy, and tissue closure
- Gynecological surgery: Hysterectomy and other pelvic procedures
Surgical staplers come in two main types: internal (endoscopic or laparoscopic) staplers used inside the body and skin staplers used to close external wounds. The most serious injuries are associated with internal staplers that malfunction during operations on vital organs and blood vessels.
How Do Surgical Stapler Injuries Happen?
Surgical stapler injuries occur when the device fails to perform as designed during surgery. The most common types of malfunctions include:
- Misfiring: The stapler fires incorrectly, placing staples at the wrong angle, depth, or spacing, resulting in tissue that is not properly sealed
- Failure to fire: The stapler jams or fails to deploy staples, forcing the surgeon to improvise during a live procedure
- Incomplete staple formation: Staples do not fully close (malform), leaving gaps that allow blood, digestive fluids, or air to leak
- Difficulty removing the stapler: The device becomes stuck in tissue and cannot be extracted without causing additional damage
- Defective staple cartridges: Cartridges that are manufactured incorrectly or loaded with the wrong size staples for the tissue being sealed
When a stapler malfunctions, the consequences during live surgery can be severe. The surgeon must address the failure in real time, often leading to extended operations, emergency interventions, and additional surgical procedures.
Injuries and Complications From Stapler Malfunctions
Patients who suffer surgical stapler injuries may experience:
- Internal bleeding: Improperly formed staple lines fail to seal blood vessels, causing hemorrhaging
- Anastomotic leaks: Connections between bowel segments fail, allowing digestive contents to leak into the abdominal cavity, causing peritonitis (a life-threatening infection)
- Organ damage: Misfired staples can puncture or damage nearby organs
- Sepsis: Bacterial infections from leaking staple lines can enter the bloodstream and become life-threatening
- Reoperation: Additional surgeries to repair damage, remove faulty staples, or address complications
- Extended hospital stays: Weeks or months of additional hospitalization for complications
- Permanent injury or disability: Some stapler injuries result in permanent bowel damage, ostomy bags, or other lasting medical conditions
- Death: In the most severe cases, stapler malfunctions have caused fatal hemorrhaging or infections
The FDA has received over 100,000 adverse event reports related to surgical staplers, including thousands of injuries and hundreds of deaths. These numbers prompted the FDA to reclassify internal surgical staplers from Class I (lowest risk) to Class II (moderate risk) in 2020, requiring manufacturers to submit additional safety data.
Which Manufacturers Are Involved?
The surgical stapler market is dominated by a few large manufacturers. The companies most frequently named in lawsuits include:
- Ethicon (Johnson and Johnson): Manufactures the Echelon, Endo-Surgery, and Proximate stapler lines. Ethicon is the most frequently sued manufacturer, with thousands of adverse event reports linked to its products.
- Medtronic (Covidien): Manufactures the Endo GIA, iDrive, and Signia stapler systems
- Intuitive Surgical: Manufactures stapling devices used with the da Vinci robotic surgical system
- Purple Surgical and other smaller manufacturers: Several smaller companies produce surgical staplers that have also been linked to adverse events
Internal documents obtained during litigation have revealed that some manufacturers were aware of elevated malfunction rates but continued to sell and distribute their devices without adequate warnings to surgeons and hospitals.
Legal Theories in Surgical Stapler Lawsuits
Surgical stapler lawsuits are filed under product liability law. Three main legal theories apply:
- Design defect: The stapler’s design was inherently flawed, making it prone to misfiring, jamming, or malforming staples under conditions that surgeons would commonly encounter
- Manufacturing defect: A specific stapler or staple cartridge deviated from design specifications during production, creating a device that was more likely to fail
- Failure to warn: The manufacturer knew about elevated malfunction rates or specific risks associated with their product but did not adequately warn surgeons, hospitals, or patients
Many lawsuits also allege that manufacturers made misleading claims about the reliability and safety of their products in marketing materials and sales presentations to surgical teams.
The FDA’s Response to Surgical Stapler Problems
The FDA has taken several actions in response to the high volume of adverse event reports associated with surgical staplers:
- 2019 Safety Communication: The FDA issued a letter to healthcare providers highlighting the risks associated with surgical staplers and staples, noting that many adverse events appeared to be related to device malfunctions rather than user error
- 2020 Reclassification: The FDA reclassified internal surgical staplers from Class I (general controls only) to Class II (special controls), requiring manufacturers to submit premarket notifications (510(k)) with safety and performance data. Previously, these devices could enter the market with minimal regulatory review.
- Enhanced reporting requirements: Manufacturers are now required to submit more detailed adverse event reports and follow updated performance standards
- Ongoing surveillance: The FDA continues to monitor stapler-related adverse events and has stated that it will take additional action if warranted
The reclassification was significant because it acknowledged that the previous regulatory framework was insufficient to protect patients. For decades, surgical staplers were treated as low-risk devices despite being used in life-or-death surgical situations. The change means that new surgical staplers entering the market must now demonstrate their safety before they can be sold.
Ethicon Lawsuits and the MDL
Ethicon (a Johnson and Johnson subsidiary) is the defendant in the majority of surgical stapler lawsuits. In 2021, federal cases against Ethicon were consolidated into a multidistrict litigation (MDL No. 2985) in the Southern District of Ohio.
The MDL process groups related cases together for pretrial proceedings, including discovery (the exchange of evidence), expert witness challenges, and bellwether trials (test cases). Key allegations in the Ethicon MDL include:
- Ethicon’s staplers had significantly higher malfunction rates than reported
- The company received thousands of internal complaints about stapler failures but did not adequately redesign the devices or warn surgeons
- Ethicon’s marketing materials overstated the safety and reliability of its products
- The company’s quality control processes were insufficient to catch manufacturing defects before devices reached operating rooms
The MDL is ongoing, with bellwether trial selections underway. The outcomes of these initial trials will significantly influence settlement negotiations and the future direction of the litigation.
Request a free consultation with Counsel Hound to discuss the legal theories that apply to your surgical stapler injury.
What Compensation Is Available?
Patients injured by defective surgical staplers can seek compensation for:
- Medical expenses: Costs of reoperation, extended hospitalization, ICU care, and treatment of complications
- Future medical costs: Ongoing care, follow-up procedures, and long-term treatment for permanent injuries
- Lost wages: Income lost during extended recovery and any time away from work due to complications
- Pain and suffering: Physical pain from the injury, additional surgeries, and recovery
- Emotional distress: Psychological impact of a surgical complication, including anxiety about future medical procedures
- Loss of enjoyment of life: Impact on daily activities and quality of life due to permanent complications
- Punitive damages: If the manufacturer knowingly concealed malfunction data or failed to act on safety concerns
For information on how settlements are taxed, see our guide on personal injury settlement taxes.
Steps to Take If You Were Injured
- Get your surgical records: Request the full operative report from your surgery, including the specific stapler model, lot number, and any notes about device malfunctions. Your surgeon’s notes about what went wrong are critical evidence.
- Identify the device: Find out the manufacturer, model name, and lot number of the surgical stapler used in your procedure. Hospital supply records and the operative report should contain this information.
- Preserve evidence: If the hospital retained the defective stapler or cartridge, ask that it be preserved. Physical evidence of the malfunction is the strongest proof in a product liability case.
- Document your complications: Keep records of all follow-up surgeries, hospital stays, medical bills, and the impact on your daily life.
- Check for FDA reports: Search the FDA’s MAUDE database for adverse event reports on the same stapler model. A pattern of similar malfunctions strengthens your case.
- Consult an attorney: A personal injury attorney experienced in medical device litigation can evaluate your case and determine whether the manufacturer is liable. Most work on a contingency fee basis.
Contact Counsel Hound today for a free case evaluation. We connect patients injured by defective medical devices with vetted, experienced attorneys on a no-fee-unless-you-win basis.
Frequently Asked Questions About Surgical Stapler Lawsuits
How do I know if my injury was caused by the stapler and not the surgeon?
This is a common question, and it is one your attorney and medical experts will investigate. If the operative report documents a device malfunction (misfire, jam, or malformed staples), the case points toward a product defect. If the FDA’s MAUDE database shows a pattern of similar malfunctions with the same device, that further supports a product liability claim rather than surgical error.
Is there a deadline to file a surgical stapler lawsuit?
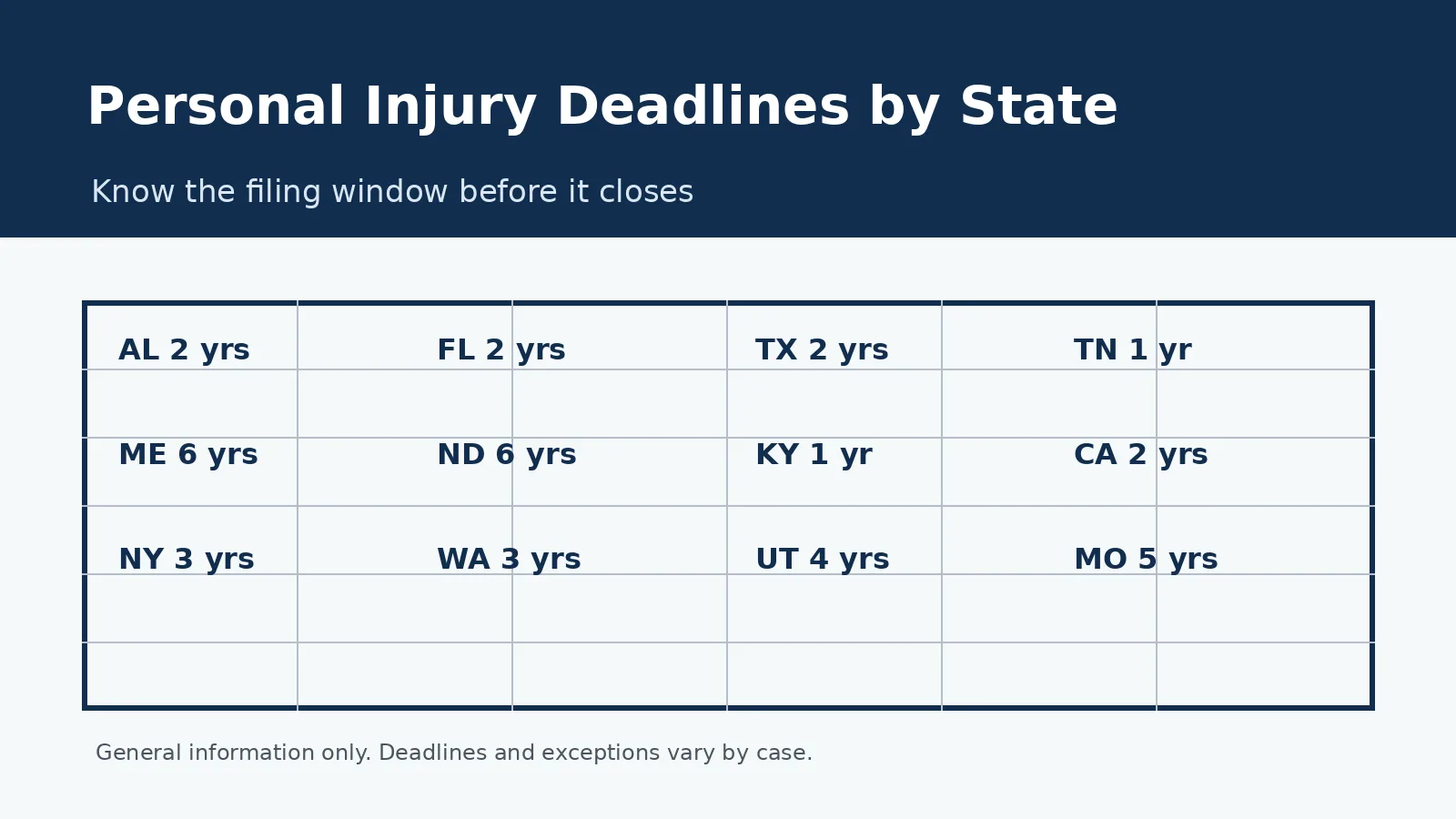
Yes. The statute of limitations varies by state, typically two to four years from the date of the injury or the date you discovered (or should have discovered) the connection between your complications and the device. Filing promptly protects your rights.
Can I sue even if my surgery was years ago?
It depends on when you learned that a stapler malfunction contributed to your complications. The discovery rule in many states starts the clock when you knew or should have known about the defect, not necessarily when the surgery occurred. An attorney can determine whether your claim is timely.
Are there class actions for surgical stapler injuries?
Some surgical stapler cases have been consolidated into multidistrict litigation (MDL) for pretrial proceedings. Individual cases within an MDL are resolved separately based on their specific facts. Your attorney will advise whether your case should be filed as part of an existing MDL or as a standalone lawsuit.
How long does a surgical stapler lawsuit take?
Timelines vary based on the complexity of the case and whether it is resolved through settlement or trial. Many medical device cases take one to three years. Cases consolidated in an MDL may follow a different schedule based on bellwether trial outcomes and settlement discussions.