Transvaginal mesh implants were once promoted as a safe treatment for pelvic organ prolapse and stress urinary incontinence. But thousands of women discovered that these defective medical devices caused serious complications, including chronic pain, infections, organ damage, and the need for additional surgeries. If you received a transvaginal mesh implant and experienced complications, you may have the right to file a lawsuit and recover compensation.
Contact Counsel Hound for a free case evaluation to find out if you qualify for a transvaginal mesh claim. There are no fees unless you win.
This guide covers the history of transvaginal mesh litigation, the complications associated with these devices, who can file a lawsuit, what compensation is available, and the steps to take if you have been affected.
What Is Transvaginal Mesh?
Transvaginal mesh is a synthetic surgical implant made of polypropylene plastic. Surgeons placed it through the vaginal wall to support weakened pelvic tissue in women diagnosed with pelvic organ prolapse (when organs like the bladder, uterus, or rectum drop from their normal position) or stress urinary incontinence (involuntary urine leakage during physical activity).
The mesh was designed to act as a permanent scaffold, reinforcing the pelvic floor. Several manufacturers marketed these devices aggressively to surgeons, claiming they offered faster recovery times and better outcomes than traditional surgical repair. In reality, the mesh frequently caused problems that were worse than the original condition it was meant to treat.
The FDA first cleared transvaginal mesh devices for pelvic organ prolapse in 2002 using the 510(k) process, which allowed the devices to reach patients without clinical trial data. This regulatory shortcut is one reason the complications were not identified until thousands of women had already been implanted.
Traditional surgical repair for pelvic organ prolapse and stress urinary incontinence uses the patient’s own tissue or sutures to provide support. While recovery may take slightly longer, traditional repair avoids the risks associated with permanent synthetic implants. Many surgeons now recommend traditional techniques over mesh, especially after the FDA ordered manufacturers to stop selling mesh for prolapse repair in 2019.
It is important to note that sling procedures for stress urinary incontinence (which use a narrow strip of mesh) are a separate category from the full mesh devices used for prolapse repair. Mid-urethral slings remain on the market and have a different risk profile, though complications can still occur. Patients with any type of mesh implant who are experiencing problems should discuss their options with a doctor and an attorney.
Complications From Transvaginal Mesh
Women with transvaginal mesh implants have reported a wide range of serious complications, many of which appear months or years after the initial surgery:
- Mesh erosion: The mesh wears through the vaginal wall, exposing sharp edges that can injure surrounding tissue and cause pain during intercourse
- Chronic pelvic pain: Persistent pain that does not respond to medication or physical therapy, often caused by the mesh contracting or adhering to nerves and tissue
- Infection: The synthetic material can harbor bacteria, leading to recurring infections that are difficult to treat while the mesh remains in place
- Organ perforation: The mesh can migrate and pierce the bladder, bowel, or other organs, requiring emergency surgery
- Urinary problems: New or worsened incontinence, difficulty urinating, and urinary tract infections
- Pain during intercourse: Sharp mesh edges or mesh contraction can make sexual activity painful or impossible
- Vaginal scarring: Scar tissue formation around the mesh causes tightening and shortening of the vaginal canal
- Mesh contraction (shrinkage): The mesh can shrink after implantation, pulling surrounding tissue and causing pain and organ displacement
- Autoimmune reactions: Some patients develop systemic inflammatory responses to the polypropylene material
Many of these complications require revision surgery to remove part or all of the mesh. Mesh removal is a complex and risky procedure because the material can become embedded in tissue, making complete removal difficult or impossible without causing additional damage.
The History of Mesh Litigation
Transvaginal mesh litigation is one of the largest mass tort actions in U.S. history. Key developments include:
- 2008: The FDA issued a Public Health Notification warning of complications associated with transvaginal mesh for pelvic organ prolapse repair
- 2011: The FDA issued an updated Safety Communication stating that serious complications from mesh use for pelvic organ prolapse are “not rare” and questioning whether mesh offered any advantage over traditional surgery
- 2012-2016: Federal courts consolidated tens of thousands of mesh lawsuits into several multidistrict litigations (MDLs), with separate MDLs for each manufacturer (Boston Scientific, C.R. Bard, Ethicon/Johnson and Johnson, Coloplast, Cook Medical, and others)
- 2016: The FDA reclassified mesh for pelvic organ prolapse from Class II to Class III, requiring manufacturers to submit premarket approval applications with clinical data
- 2019: The FDA ordered manufacturers to stop selling and distributing mesh for pelvic organ prolapse after they failed to submit the required safety data
Billions of dollars in settlements and verdicts have been awarded to mesh victims. Boston Scientific alone has paid over $2.5 billion to settle mesh claims. Other manufacturers, including Ethicon, C.R. Bard, and Coloplast, have also paid substantial settlements.
Who Can File a Transvaginal Mesh Lawsuit?
You may qualify to file a transvaginal mesh lawsuit if:
- You had a transvaginal mesh implant for pelvic organ prolapse or stress urinary incontinence
- You experienced complications such as mesh erosion, chronic pain, infection, organ damage, or the need for revision surgery
- Your complications are linked to the mesh implant rather than the surgical procedure itself
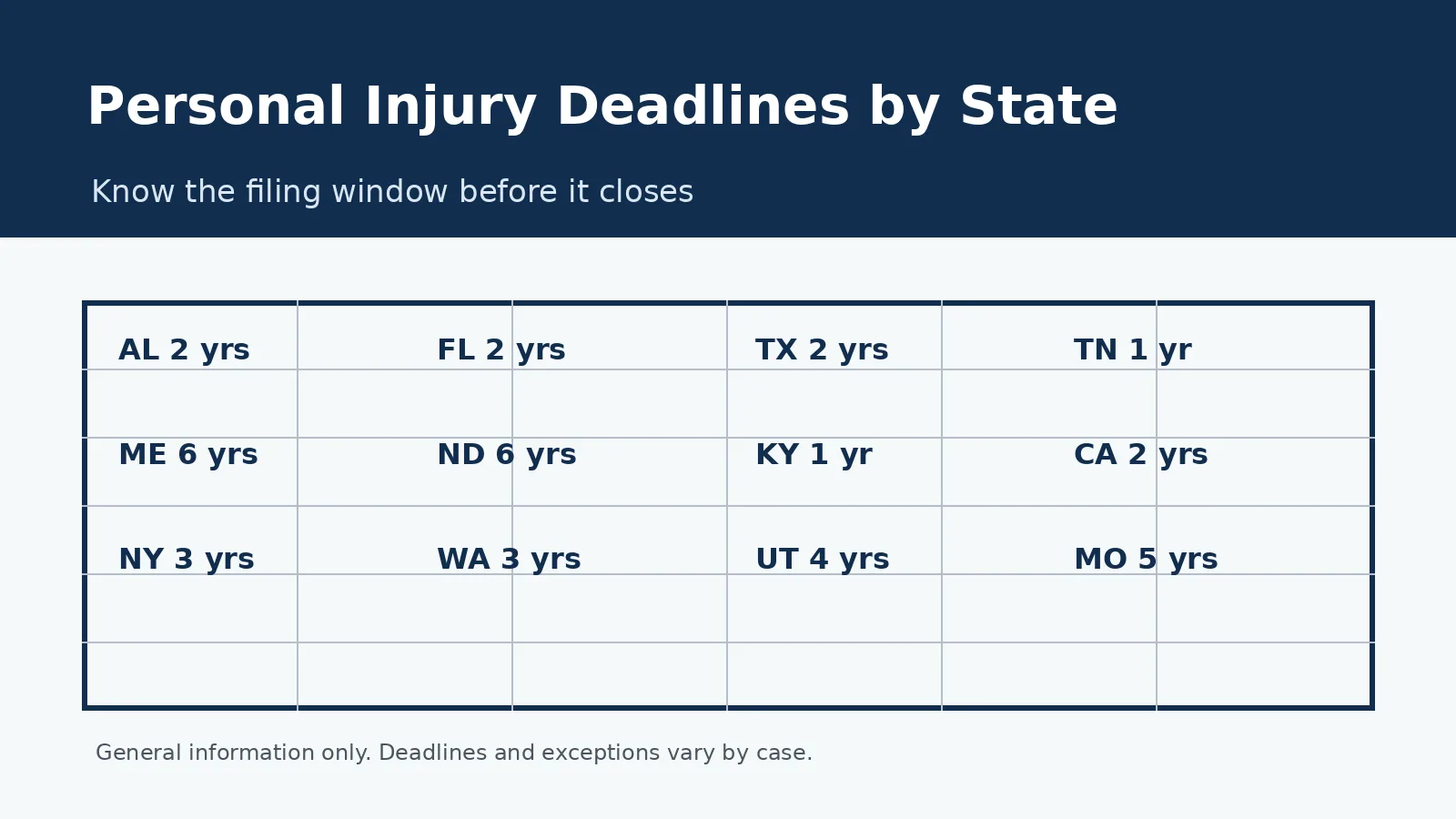
Even if you had your mesh implanted years ago, you may still have a valid claim. Many complications do not appear until months or years after the surgery. The statute of limitations in most states starts from the date you first experienced symptoms or learned about the connection between your problems and the mesh, not the date of the original implant surgery.
Family members and spouses may also have claims. Loss of consortium claims allow spouses to seek compensation for the impact of mesh complications on their relationship, including lost companionship and intimacy.
Request a free consultation with Counsel Hound to discuss whether you qualify for a transvaginal mesh claim.
Legal Theories in Mesh Lawsuits
Transvaginal mesh lawsuits are built on several legal theories that hold manufacturers accountable for the harm their products caused:
- Design defect: The mesh was inherently dangerous because of its design. The polypropylene material was not suitable for permanent implantation in the human body because it degrades, contracts, and provokes inflammatory responses over time. Even when manufactured exactly as designed, the product was unreasonably dangerous.
- Manufacturing defect: Some individual mesh devices deviated from the manufacturer’s specifications during production, resulting in products that were even more prone to failure.
- Failure to warn: Manufacturers knew or should have known about the risks of mesh erosion, chronic pain, and other complications but failed to adequately warn doctors and patients. Internal company documents obtained during litigation have revealed that some manufacturers were aware of high complication rates before the devices reached the market.
- Negligence: Manufacturers failed to exercise reasonable care in testing, marketing, and monitoring their products. Rushing devices to market without clinical trials and failing to act on early reports of complications are examples of negligent conduct.
- Breach of warranty: Manufacturers made express or implied promises about the safety and effectiveness of their mesh products that turned out to be false.
What Compensation Is Available?
Transvaginal mesh lawsuit settlements and verdicts compensate patients for the full impact of the device’s failure:
- Medical expenses: Costs of treating mesh complications, including revision surgery, hospitalization, pain management, and ongoing care
- Future medical costs: Projected expenses for continued treatment, follow-up surgeries, and long-term care
- Lost wages: Income lost during recovery from complications and revision surgeries
- Pain and suffering: Physical pain from mesh erosion, contraction, and related complications
- Emotional distress: Anxiety, depression, and the psychological toll of living with chronic pain and intimate health problems
- Loss of enjoyment of life: Inability to exercise, travel, or participate in activities due to pain and physical limitations
- Loss of consortium: Impact on marital and intimate relationships
- Punitive damages: Additional damages in cases where manufacturers knew about risks and failed to warn patients or doctors
Individual settlement amounts in mesh cases have ranged from $50,000 to over $1 million, depending on the severity of complications, the number of revision surgeries, and the strength of the evidence. For information about how settlements are taxed, see our guide on personal injury settlement taxes.
Steps to Take If You Have a Mesh Implant
Whether you are currently experiencing complications or want to understand your options, here is what to do:
- Document your symptoms: Keep a detailed record of pain levels, bleeding, infections, urinary problems, and any other symptoms you experience. Note when they started and how they affect your daily life.
- See your doctor: Get a thorough examination to determine whether your symptoms are related to the mesh implant. Ask your doctor to document the findings in your medical records and specify any connection to the mesh.
- Identify your implant: Find out the manufacturer, brand name, and model of your mesh implant. Your surgeon’s office should have this information in your operative report.
- Gather medical records: Collect records from the original surgery, follow-up visits, and any treatment for complications. Include operative reports, imaging results, and pathology reports if mesh was removed.
- Do not delay: Statute of limitations deadlines vary by state. Speaking with an attorney as soon as possible protects your right to file, even if you are unsure whether you have a valid claim.
- Consult a personal injury attorney: An experienced attorney can evaluate your case, identify the manufacturer, and handle the legal process. Most mesh attorneys work on a contingency fee basis, so you pay nothing unless you recover compensation.
Contact Counsel Hound today for a free case evaluation. We connect women affected by mesh complications with vetted, experienced attorneys who handle medical device lawsuits on a no-fee-unless-you-win basis.
Frequently Asked Questions About Transvaginal Mesh Lawsuits
Is it too late to file a transvaginal mesh lawsuit?
It depends on your state’s statute of limitations and when you first experienced or became aware of mesh-related complications. Many states apply a “discovery rule” that starts the clock when you knew or should have known about the connection between your symptoms and the mesh, not when the surgery was performed. An attorney can evaluate whether your claim is still within the filing deadline.
Can I file a lawsuit if my mesh has not been removed yet?
Yes. You do not need to have had revision surgery to file a lawsuit. If you are experiencing complications from a mesh implant, you may have a valid claim even if the mesh is still in place. However, documenting your symptoms and getting a medical evaluation is important for building your case.
What is the difference between a mesh lawsuit and a class action?
Most transvaginal mesh cases are handled through multidistrict litigation (MDL), not class actions. In an MDL, each plaintiff maintains their own individual case with their own facts and damages. The MDL process groups cases together for shared pretrial proceedings to improve efficiency, but each case is resolved individually based on its specific merits.
What if my mesh was for a hernia, not pelvic repair?
Hernia mesh lawsuits are a separate but related area of litigation. Many of the same manufacturers (including C.R. Bard, Ethicon, and Atrium) face lawsuits over defective hernia mesh products that caused infections, adhesions, and bowel obstructions. If you had hernia mesh complications, you may also have a valid claim.
How long does a transvaginal mesh lawsuit take?
Timelines vary depending on the specific manufacturer, the court, and whether the case settles or goes to trial. Some cases in the larger MDLs have settled within one to two years after filing, while others take longer. Your attorney will provide updates throughout the process and work to resolve your case as efficiently as possible.